RWE in China is moving beyond pilots and into pricing decisions. With new NHSA frameworks and State Council Guidance linking RWE to NRDL access and potential price upside, the role of evidence is shifting rapidly. What does this mean for manufacturers?
RWE has come a long way in China
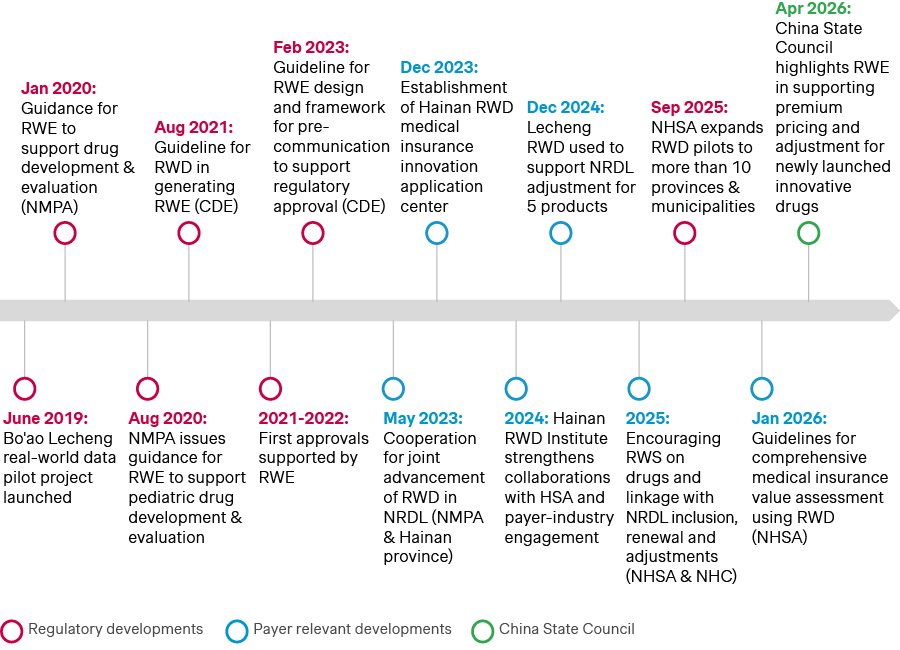
RWE has been gaining increasing relevance in China since the first pilot launched alongside the Hainan Bo’ao early access program in 2019.
Source: Simon-Kucher insights; CDE: Center for Drug Evaluation; HSA: Health Security Administration; NHC: National Health Commission; NHSA: National Healthcare Security Administration; NMPA: National Medical Product Administration; NRDL: National Reimbursement Drug List; RWD: Real-world data; RWE: Real-world evidence.
In 2020, China’s regulatory authority, the National Medical Products Administration (NMPA) issued the first guidance on RWE to support drug development and approval. Since then, innovative therapies like Gavreto, Sarclisa, Cosela, and Leqvio have leveraged RWE data from the Bo’ao pilot to support accelerated China approvals.
Subsequently, RWE has also been increasingly adopted in National Reimbursement Drug List (NRDL) decision-making. Since 2023, RWE has shifted from a largely exploratory concept to a policy-supported tool for NRDL, with increasing applications through structured programs.
By 2025, National Healthcare Security Administration (NHSA) expanded these efforts beyond a single pilot to multiple regions nationwide, while advancing methodological research and application standards to support more systematic use of RWE in NRDL evaluations. Today, RWE is playing an increasingly tangible role in China market access. Most recently, Sarclisa and Leqvio secured NRDL listings, while Revestive and Yescarta were included in the first C-list in 2025 thanks in no small part to the real-world data they furnished.
| Product | Company | Regulatory | NRDL | C-list | RWE design |
| Sarclisa (isatuximab) | Sanofi | Close evidence gaps, mitigating key global trial w/o CN data | Close evidence gap on efficacy, safety & QoL profile | N/A |
|
| Gavreto (pralsetinib) | CStone | Close evidence gaps, mitigating single-arm Ph1/2 trial | N/A | N/A |
|
| Cosela (trilaciclib) | Simcere | Close evidence gaps, validating the protective effect of trilaciclib on CIM | Close evidence gaps, efficacy vs. comparator | N/A |
|
| Leqvio (inclisiran) | Novartis | Close evidence gaps, validating durable lipid-lowering efficacy in CN | Close evidence gaps, local evidence vs. PCSK9 mAbs Address payer concerns, label-aligned dosing in China | N/A |
|
| Kaitanni (cadonilimab) | Akesobio | N/A | Close evidence gaps, safety under label-aligned use | N/A |
|
| Sotyktu (deucravacitinib) | BMS | N/A | Close evidence gaps on Asian population evidence | N/A |
|
| Tabrecta (capmatinib) | Novartis | N/A | Close evidence gaps, mitigating single-arm clinical trial | N/A |
|
| Orpathys (savolitinib) | AstraZeneca | N/A | Close evidence gaps, mitigating single-arm clinical trial | N/A |
|
| Revestive (teduglutide) | Takeda | N/A | N/A | Close evidence gaps, mitigating key global trial w/o CN data |
|
| Yescarta (axicabtagene ciloleucel) | FosunKite | Close evidence gaps, mitigating key global trial w/o CN data | N/A | Close evidence gaps, mitigating key global trial w/o CN data |
|
Source: Simon-Kucher insights; 2025 NRDL dossier.
The new paradigm for China RWE
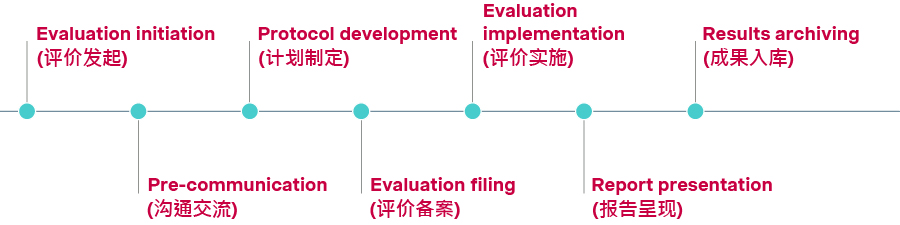
In January 2026, NHSA issued a set of policies on adopting RWE for comprehensive NRDL value assessment, outlining process steps, peer-review requirements, and standards for effectiveness, safety, and pharmaco-economics.
For the first time, a seven-step process has been laid out, with detailed guidance for manufacturers. The framework places an overarching emphasis on procedural rigor, quality, and transparency. Compared with many overseas HTA systems, China’s approach highlighted in particular the importance of upfront protocol communication, commitment to publish results, and close coordination throughout the evidence generation and evaluation process.
Source: Simon-Kucher insights; NHSA.
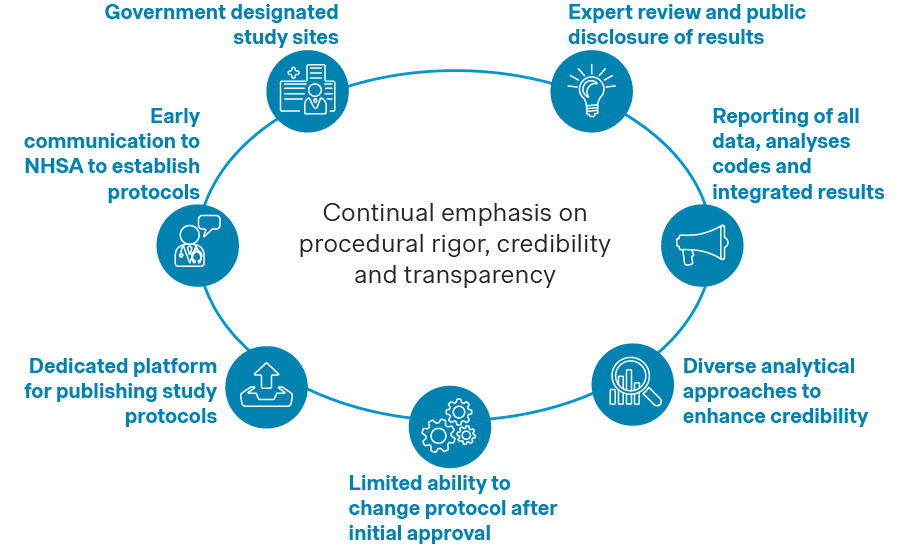
Manufacturers are encouraged to include designated RWE evaluation sites for their real-world studies, share protocols with NHSA beforehand, and publish qualification information and study protocols on designated platforms. Statistical analysis plans should be aligned ahead of time, with multiple analytical approaches applied to ensure data quality. Integrated results are expected to clearly address pre-specified research questions, and manufacturers should also prepare for expert review and public disclosure.
Source: Simon-Kucher insights.
More recently, the State Council Guidance on Drug Pricing issued in April 2026 once again put RWE in the spotlight as a basis for premium pricing and even upward adjustments for newly launched innovative drugs, reinforcing its expanding role in China.
Largely building upon existing policies
| Topic | Key contents | Near term actionability |
Launch price formation mechanism (新上市药品首发价格机制) |
|  |
RWE generation to support new launch pricing and NRDL (药品真实世界研究框架) |
|  |
Involvement of multiple stakeholders for funding of innovative drugs (促进创新药多元支付) |
|  |
VoBP implementation (药品集中带量采购价格形成机制) |
|  |
Source: Simon-Kucher insights. BMI: Basic Medical Insurance; CHI: Commercial health insurance; NRDL: National Reimbursement Drug List; RWE: Real-world evidence; VoBP: Volume-based procurement.
RWE for China access
NHSA acknowledges that RWE can generate critical data and evidence toward key NRDL value dimensions, including demonstrating disease burden and unmet needs, showcasing clinical, economic, and equity value, and addressing safety and budget impact concerns.
As NHSA formalizes RWE processes and the State Council issues new pricing guidance, manufacturers should plan early to:
- Demonstrate relevance to China’s unique treatment realities
- Quantify China-specific disease burden and patient characteristics. This includes treated prevalence, disease severity, and special populations. It should also reflect diagnostic hurdles, differences in care patterns across centers of excellence vs. tertiary centers, or comorbidities with varying prevalence in China compared to other countries.
- Assess local treatment patterns, standard of care, and appropriateness of proposed comparator, as treatment paradigms in China may differ significantly from other markets given the rapid growth of the domestic biopharma industry and the availability of China-specific innovative therapies.
- Highlight critical unmet needs by identifying where current care falls short – for example, due to fragmented treatment pathways, high discontinuation rates, or inconsistent clinical practices.
- Close evidence gaps that matter
- Substantiate real-world effectiveness and safety, particularly for products approved on the basis of limited evidence, such as single-arm studies, surrogate endpoints, or shorter trials.
- Demonstrate effectiveness in Chinese patients not sufficiently represented in global pivotal trials.
- Address evidence gaps in under-represented patients, particularly in the pediatric population, those with major comorbidities or polypharmacy, or those treated outside of typical care settings.
- Address payer concerns
- Strengthen payer confidence in budget impact estimates by using RWE to inform real-world dosing and treatment duration, particularly where dosing is expected to be highly variable or complex. RWE can also help define the actual eligible patient population and estimate uptake and market share in the Chinese clinical practice.
- Capture the full value of treatment beyond direct medical costs by using RWE to show reductions in healthcare resource utilization, such as fewer hospital visits, shorter lengths of stay, and higher productivity gains for patients and caregivers, strengthening the broader value narrative.
Translating RWE into access advantage
The path toward broader use of RWE in access decision-making will hinge on the quality of the evidence and methodologies. Following the January 2026 guidance, NHSA signed agreements with 79 leading hospitals to strengthen RWE standards, data governance, and pilot implementation.
In that context, companies that proactively invest in a robust RWE strategy, trusted partnerships, and well-planned real-world studies will be best positioned to leverage these developments going forward. From our experience, it would be key to:
- Start early and build RWE into China strategy from the outset. Companies should identify whether and where RWE could strengthen the NRDL strategy and plan for it well before NRDL engagement, and in some cases even before NMPA approval.
- Prioritize evidence gaps that matter for reimbursement. Focus on topics most relevant to NRDL and C-list decision-making, including China-specific effectiveness and safety data, healthcare resource utilization costs, and budget impact considerations.
- Plan designs in line with NHSA requirements. As the process is now more formalized, companies need to ensure their study goals and protocols are well aligned to meet the new standards.
- Build trusted partnerships early. With NHSA expanding pilots to designated hospitals and evaluation centers, companies should invest early in relationships with qualified partners that can support high-quality RWE generation.
- Map out actionable engagement plans. Pursue proactive engagements with key stakeholders across the access ecosystem, take part in and take the lead on RWE initiatives, and position early to influence and shape the changes going forward.
With contributions from Mingyue Gao, Keshi Li, and Yiwen Ying.




