For about a year and a half, doctors in Germany have been able to prescribe digital health applications (DiGAs), which are reimbursed by statutory health insurers. For reimbursement, DiGA manufacturers and the German National Association of Statutory Health Insurance Funds (GKV-SV) must negotiate a price. If no agreement is reached, an arbitration board sets the reimbursement price. How can DiGA manufacturers prepare for their price negotiations? Here are four key learnings from the first DiGA price negotiations.
Since October 2020, doctors have been able to prescribe digital health applications (DiGAs) that are officially approved by the Federal Institute for Drugs and Medical Devices (BfArM), and reimbursed by the statutory health insurance funds (GKV) in Germany.
Until now, DiGA manufacturers set their own prices during the first year of reimbursement. Starting from the 13th month, the price that applies is the one negotiated by the DiGA manufacturer and the GKV-SV.
Negotiations between DiGA manufacturers and the GKV-SV: First price decisions made by the arbitration board
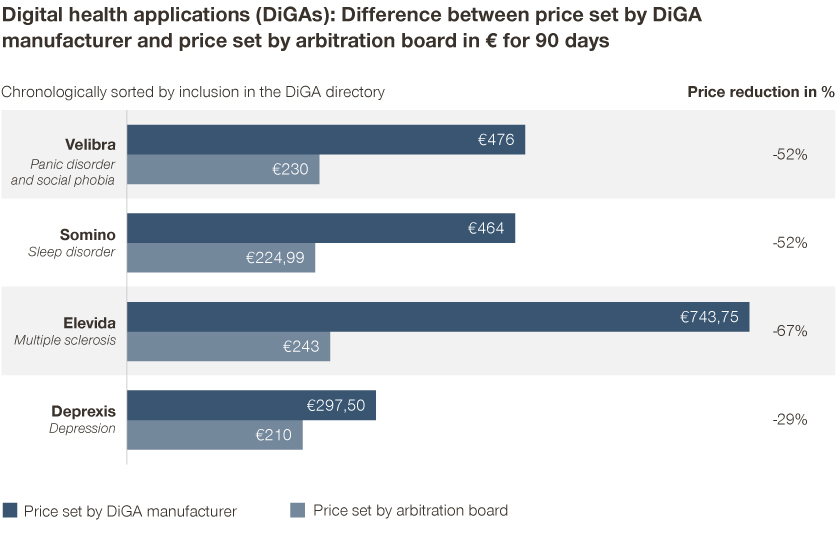
The first four negotiations between DiGA manufacturers and the GKV-SV up to today were unsuccessful. In all four cases– three apps from the mental health category and one from the nervous system category – the arbitration board set the reimbursement prices. The majority of manufacturers had to accept considerable price reductions.
The new prices for a 90-day access period range from 210 to 243 euros. On average, DiGA manufacturers have had to cut their first-year prices by half.
So, how can DiGA manufacturers best prepare for price negotiations?
The following four learnings from the first DiGA price negotiations provide insights.
Price negotiations with the GKV-SV: Four recommendations for DiGA manufacturers
1. Develop your evidence strategy in line with your future price negotiation strategy
Evidence is becoming increasingly relevant for reimbursement/pricing strategies. The past DiGA price negotiations proved that price is largely determined by the extent of the positive healthcare effects. In practice, a distinction is made between the magnitude of the positive healthcare effects demonstrated and the quality of the studies submitted as evidence.
Quality is more important than quantity. Manufacturers do better by conducting and submitting fewer but higher-quality studies. Their focus should therefore be on a strong study design in line with the intended use of the DiGA as well as large effect sizes.
Manufacturers may also consider generating additional evidence to strengthen their bargaining power. For example, with the growing emergence of apps in the DiGA market, they could conduct studies comparing their app with DiGAs for the same indication.
Therefore, preparing the price negotiations begins with planning studies and generating price-substantiating evidence. During the price negotiation, they should present a strong body of evidence that can serve both dimensions – the magnitude of healthcare effects and quality of evidence.
2. Set the right start price
When setting their launch price, DiGA manufacturers should proceed strategically, as the GKV-SV and arbitration board consider the actual price in their pricing decision (in addition to the demonstrated positive healthcare effects).
While manufacturers have been able to freely set their start price in the first DiGA negotiations, the introduction of maximum price limits (“Höchstbeträge”) will limit the scope for adjusting prices in the future. There is still more leeway for DiGAs in categories where maximum price limits do not apply yet, but there will also be a cap in the long term.
If DiGA manufacturers cannot claim an exemption, they must be prepared for their start price to be reduced to or below the price cap.
In addition, information such as out-of-pocket prices including prices for privately insured persons or prices in other European countries must be disclosed during the BfArM application process.
It’s therefore essential that manufacturers thoroughly evaluate how they will set their launch price and negotiate other price agreements.
3. Know your price anchor
To justify the asking price, manufacturers and the GKV-SV reference comparable therapy options in healthcare that are in accordance to the DiGA and its area of application. These serve as reference point for the monetization potential of the DiGA’s benefits.
Comparable healthcare costs play a particularly important role for the GKV-SV when determining the reimbursement amount. The GKV healthcare costs for psychotherapy formed the starting point in the three completed price negotiations of the mental health DiGAs. More specifically, the costs of psychotherapy in a group setting with nine participants were considered.
DiGA manufacturers must therefore identify a suitable anchor for pricing when defining their negotiation strategy and leverage it optimally. They also need to consider that there may already be a clear calculation model for the monetary valuation of the price anchor. This is the case for mental health DiGAs according to the existing arbitration decisions.
4. Develop an optimal negotiation strategy and tactics
- Start preparing for negotiations during the BfArM application process
The price negotiation strategy should be set long before the first negotiation round. Setting the strategic direction before submitting the application to the BfArM is important since the GKV-SV will take this information into account when setting prices.
DiGA manufacturers should ensure that their positioning and the evidence strategy they submit in the application process is consistent with their future price negotiation strategy. Information published in the DiGA directory containing statements that weaken the basis for argumentation in price negotiations with the GKV-SV should be avoided.
- Identify your negotiation levers and develop an overarching strategy
Furthermore, it’s important to consider all key negotiation parameters in the overarching strategy. Price anchors, evidence, positive healthcare effects, and price information should therefore be presented as a suitable umbrella in the form of an overarching strategy.
The negotiations that have already taken place indicate limited room for maneuver. For example, in the category of mental health DiGAs, it will be difficult to deviate from the arbitration board’s calculation model and justify a different price anchor than the statutory health insurance costs for psychotherapy in a group setting. In addition, freedom in price setting is limited for DiGAs where ceiling prices apply.
- Know your counterpart
- Anticipate the position of the GKV-SV: DiGA manufacturers should anticipate the position of the GKV-SV and develop an objection handler to be able to counterargue possible points of criticism. It’s important to have a clear picture of the negotiating partner: How did previous negotiations go? What kind of agreement was reached? What negotiation styles and techniques did the respective negotiator use? The GKV-SV is not an unknown negotiating partner for some manufacturers. DiGA manufacturers that have already encountered the GKV-SV in AMNOG procedures have a clear advantage.
- Anticipate the position of the arbitration board: Before manufacturers approach the arbitration board, they should conduct a risk-benefit analysis to determine whether they can achieve a better negotiation outcome than with the GKV-SV. The first DiGA negotiations provide cues into what level of reimbursement the arbitration board is willing to offer, and having this information can help in anticipating its position.
Early preparations and a strategic approach are key for price negotiations with the GKV-SV and arbitration board
It’s important that DiGA manufacturers have compelling evidence, a cleverly set launch price, a suitable price anchor, and an overarching negotiation strategy anticipating their negotiating partner’s next move. These four elements are essential for mastering price negotiations with the GKV-SV and the arbitration board. Planning ahead pays off.
Get in touch with our authors to learn more!








